Menu

Molar mass is measured in grams per mole. The mass in grams of one mole of a material is known as molar mass. What is molar mass, and what is its unit? The mass of a specimen of a chemical compound divided by the amount of material in that specimen, expressed in moles, is the molar mass of that compound.ģ. 
Add everything up and convert to grams/mole.Multiply every element’s atomic weight by the number of atoms included in the compound.Calculate the number of atoms of every element in the compound using the chemical formula.The standard unit is g mol⁻¹ and the SI one as kg mol⁻¹. 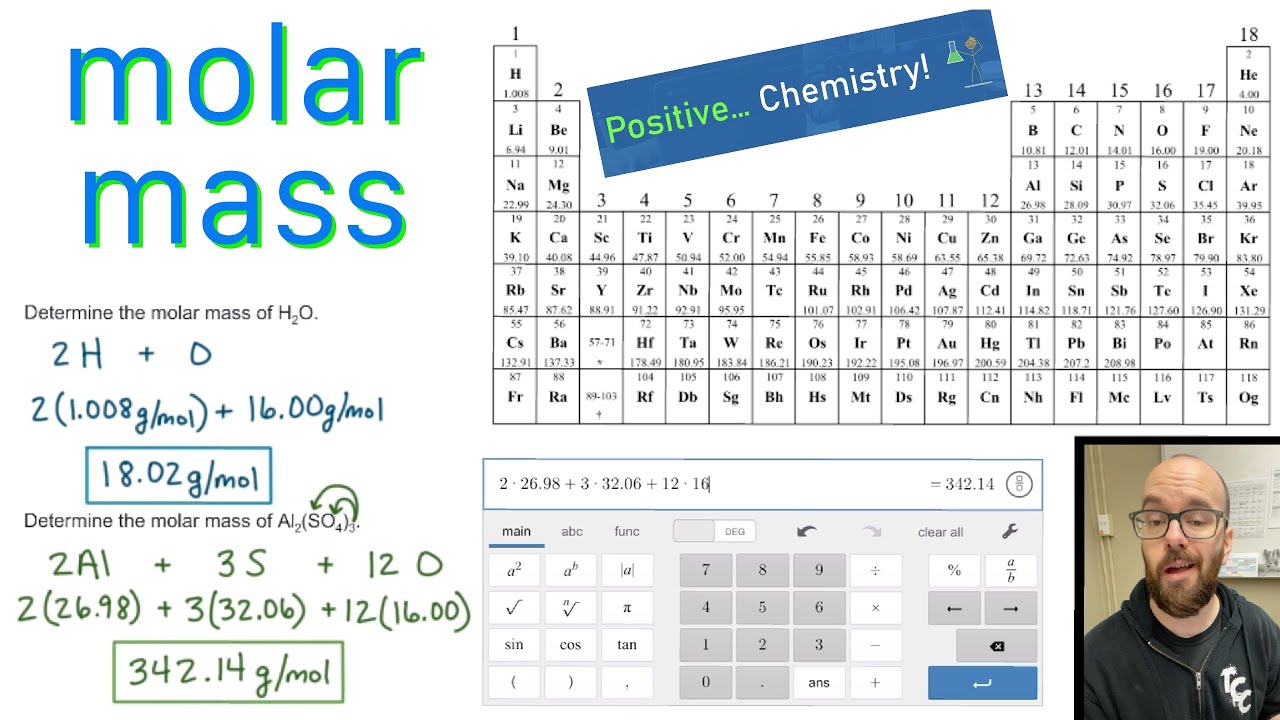
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
